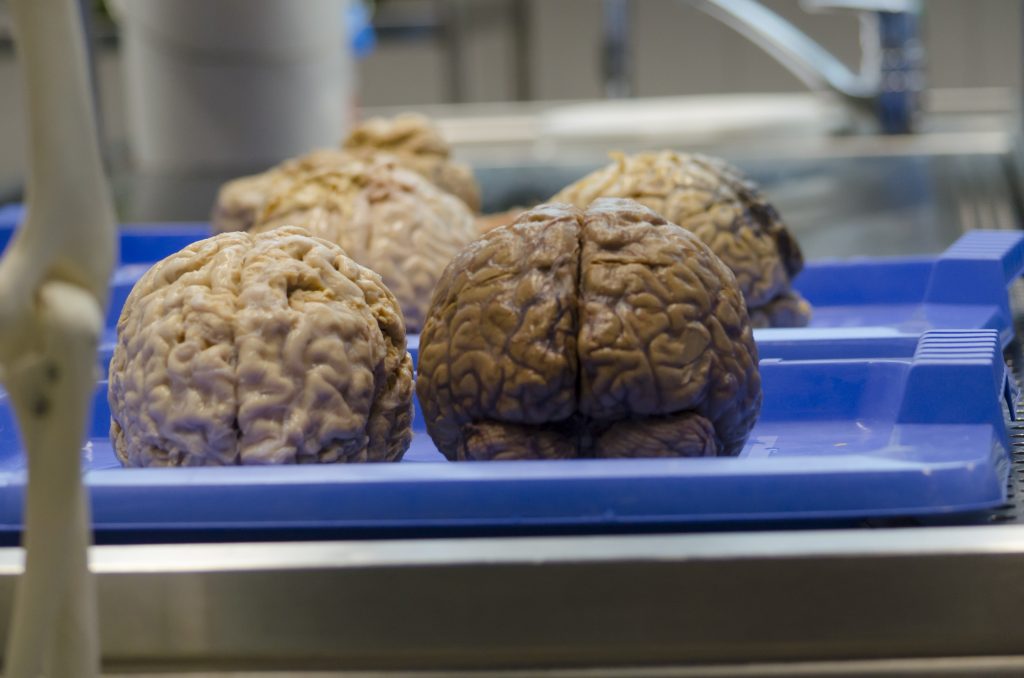
 It can be puzzling — if not outright humorous — to observe the warnings in many pharmaceutical advertisements about how a drug’s side effects can be so severe that the potential harms outweigh the possible benefits. What’s not at all funny is when one of those side effects causes a patient actual harm.
It can be puzzling — if not outright humorous — to observe the warnings in many pharmaceutical advertisements about how a drug’s side effects can be so severe that the potential harms outweigh the possible benefits. What’s not at all funny is when one of those side effects causes a patient actual harm.
Cory Jenkins began taking the FDA-approved drug Abilify in October, 2010 as part of ongoing treatment of his condition. One known side effect of Abilify is tardive dyskinesia, a serious neurological disorder that causes muscle twitching. Jenkins began showing symptoms of dyskinesia in late 2012 and early 2013. He visited the Ochsner Medical Center in New Orleans and was instructed to stop taking Abilify. Shortly thereafter the twitching ceased. By August of 2013, the symptoms returned, even though Jenkins was no longer taking Abilify. In October, 2013 Jenkins sought care from several neurologists, including one who officially diagnosed him with dyskinesia. In October, 2014 Jenkins filed a lawsuit for damages against Bristol-Myers Squibb Company and Otsuka American Pharmaceutical Inc., the makers of Abilify.
In the U.S. District Court for the Eastern District of Louisiana (“District Court”), Jenkins asserted two claims under the Louisiana Products Liability Act (LPLA). The District Court held that both claims had prescribed — meaning Jenkins did not file within the time required to commence an action — and granted the defendants’ motions for summary judgment. Claims brought under LPLA have a one-year prescriptive period; the period begins from the day the injury occurs or when damage is sustained. La. C.C. art. 3492. Damages are said to be “sustained” when they have revealed themselves with enough certainty to support the existence of a cause of action. In Louisiana, the start of the prescriptive period does not depend on a physician’s diagnosis. Instead, what controls is the date the injury occurred. Jenkins argued that there was a factual dispute over whether he had developed dyskinesia in April, and that it was not certain until his diagnosis in August. But because Jenkins admitted in his pleadings that his symptoms began in April, 2013, the District Court held that the prescriptive period for his claims against the defendants began running in April, 2013.
 Relationships between employees and employers can sour quickly when employees commit negligence during their duties. In some cases, the innocent party can seek compensation from the employer for damages caused by the employee. But what if the party causing harm is not really an employee but an independent contractor? And what if the roles are not entirely clear? A recent injury case out of Denham Springs addressed those questions.
Relationships between employees and employers can sour quickly when employees commit negligence during their duties. In some cases, the innocent party can seek compensation from the employer for damages caused by the employee. But what if the party causing harm is not really an employee but an independent contractor? And what if the roles are not entirely clear? A recent injury case out of Denham Springs addressed those questions.  Louisiana Personal Injury Lawyer Blog
Louisiana Personal Injury Lawyer Blog


 Sometimes, commonplace items such as stairs can lead to serious injuries. This case involves the unfortunate situation of a woman who fell down stairs and was injured. Under what circumstances can a building owner be held responsible for injuries from falling down the stairs?
Sometimes, commonplace items such as stairs can lead to serious injuries. This case involves the unfortunate situation of a woman who fell down stairs and was injured. Under what circumstances can a building owner be held responsible for injuries from falling down the stairs?  When you are preparing for a lawsuit, it is crucial to understand what evidence you will be allowed to present in support of your claim. On the flip side, if there is evidence you do not think the other party should be able to present, you can file a motion to try to exclude that evidence. Rulings on evidence can have a major effect on a case because they limit what a jury gets to see or hear.
When you are preparing for a lawsuit, it is crucial to understand what evidence you will be allowed to present in support of your claim. On the flip side, if there is evidence you do not think the other party should be able to present, you can file a motion to try to exclude that evidence. Rulings on evidence can have a major effect on a case because they limit what a jury gets to see or hear.  Car manufacturers have a duty to provide safe vehicles for drivers and passengers. A safe car includes an airbag that can help lessen the effects of an auto accident. What happens when your airbag doesn’t expand during a wreck? Can you sue the manufacturer of that airbag? An Ascension Parish lawsuit shows why the absence of a car that has a defective airbag will cause major headaches for your product liability lawsuit.
Car manufacturers have a duty to provide safe vehicles for drivers and passengers. A safe car includes an airbag that can help lessen the effects of an auto accident. What happens when your airbag doesn’t expand during a wreck? Can you sue the manufacturer of that airbag? An Ascension Parish lawsuit shows why the absence of a car that has a defective airbag will cause major headaches for your product liability lawsuit.  Some doors, such as bathroom doors, are heavy and have quick automatic closing mechanisms attached. If a door of that nature hits you in the back on your way and knocks you down, who is liable? The following case out of New Orleans shows how courts deal with door-closing mechanisms and trip-and-fall lawsuits.
Some doors, such as bathroom doors, are heavy and have quick automatic closing mechanisms attached. If a door of that nature hits you in the back on your way and knocks you down, who is liable? The following case out of New Orleans shows how courts deal with door-closing mechanisms and trip-and-fall lawsuits.  No one wants to experience pain and suffering after spending money and time getting surgery. Nonetheless, some people fall victim to these complications. A person can be prone to injury when a defective product is inserted into their body. This was the case with Kale Flagg.
No one wants to experience pain and suffering after spending money and time getting surgery. Nonetheless, some people fall victim to these complications. A person can be prone to injury when a defective product is inserted into their body. This was the case with Kale Flagg. Hydraulic steering is part of modern-day recreational vessels. When a boat’s hydraulic steering fails, what party bears liability? The owner, driver, or manufacturer? In the following case, the Louisiana 3
Hydraulic steering is part of modern-day recreational vessels. When a boat’s hydraulic steering fails, what party bears liability? The owner, driver, or manufacturer? In the following case, the Louisiana 3 The legal system is complicated, with many “dos-and don’ts.” Whether or not you can have your case heard in court first requires following the rules guiding the sufficiency of your claim. If your complaint fails to show that you have a right to bring the case against your defendant, your case might be dismissed. But how strictly interpreted is this rule? What does it look like when a cause of action is sufficient to be heard or ripe for dismissal?
The legal system is complicated, with many “dos-and don’ts.” Whether or not you can have your case heard in court first requires following the rules guiding the sufficiency of your claim. If your complaint fails to show that you have a right to bring the case against your defendant, your case might be dismissed. But how strictly interpreted is this rule? What does it look like when a cause of action is sufficient to be heard or ripe for dismissal? Imagine an injury on a ladder, lawnmower, boat, or other manufactured product. The product might appear defective; however, is defectiveness sufficient to win a lawsuit against the manufacturer? Under Louisiana law, to prevail in a lawsuit alleging medical injuries from a defective product, a plaintiff must provide adequate medical evidence to support that the injuries likely resulted from the defective product. This is referred to as “medical causation.” Without establishing medical causation, you may not be able to recover for your injuries.
Imagine an injury on a ladder, lawnmower, boat, or other manufactured product. The product might appear defective; however, is defectiveness sufficient to win a lawsuit against the manufacturer? Under Louisiana law, to prevail in a lawsuit alleging medical injuries from a defective product, a plaintiff must provide adequate medical evidence to support that the injuries likely resulted from the defective product. This is referred to as “medical causation.” Without establishing medical causation, you may not be able to recover for your injuries.  It can be puzzling — if not outright humorous — to observe the warnings in many pharmaceutical advertisements about how a drug’s side effects can be so severe that the potential harms outweigh the possible benefits. What’s not at all funny is when one of those side effects causes a patient actual harm.
It can be puzzling — if not outright humorous — to observe the warnings in many pharmaceutical advertisements about how a drug’s side effects can be so severe that the potential harms outweigh the possible benefits. What’s not at all funny is when one of those side effects causes a patient actual harm.